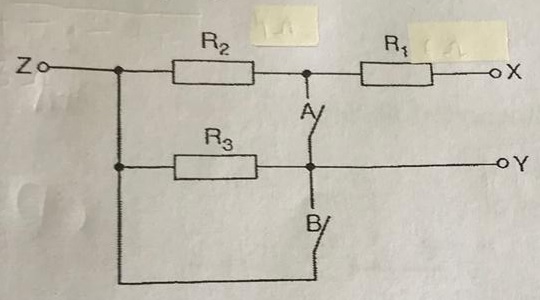
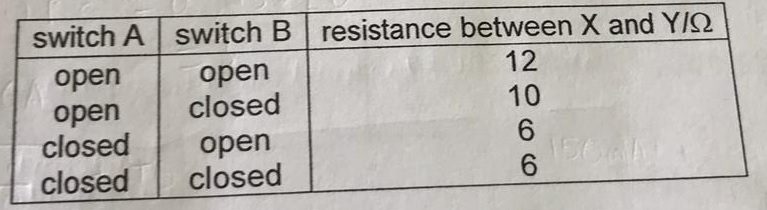
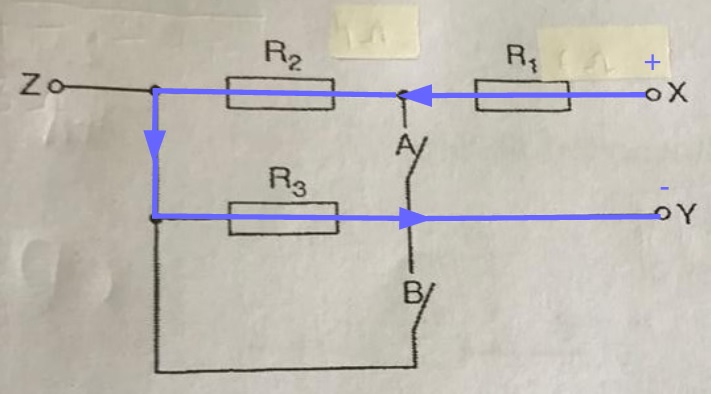
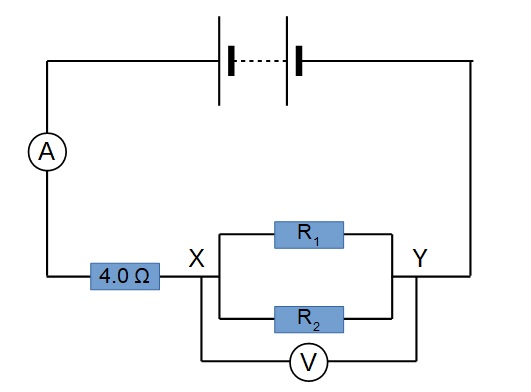
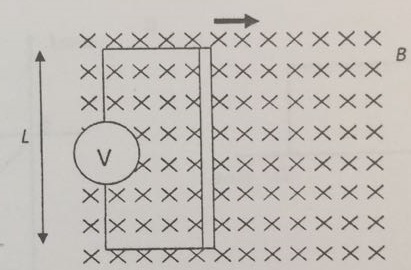
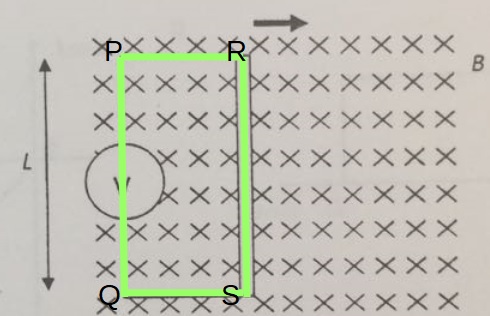
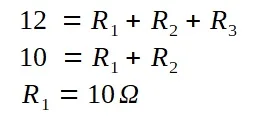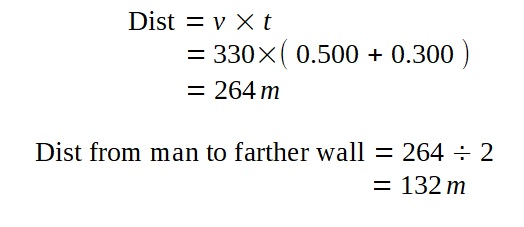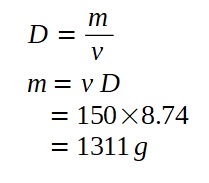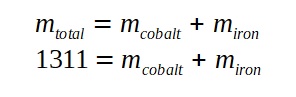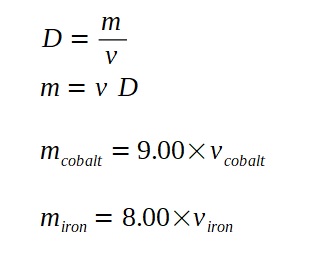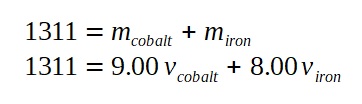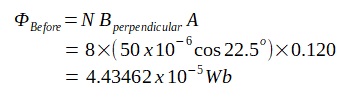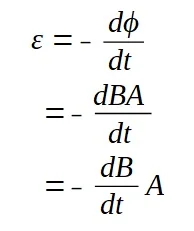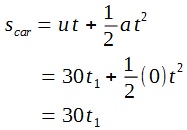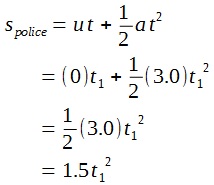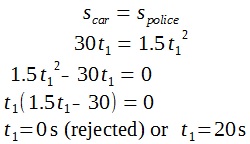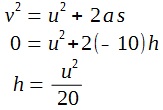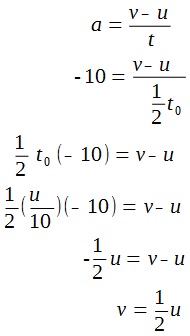A lamp is placed 30 cm above a metal surface which contains atoms of diameter 20 x 10-10 m. The lamp can be considered as a point source with power 0.015 W. It may be assumed that each electron can collect energy from a circular area which has a radius equal to that of the atom
a i) Find the intensity of the electromagnetic radiation directed on the atom.
ii) Hence, calculate the power incident on each atom.
iii) Determine, on the basis of wave theory, the time required for an electron to collect sufficient energy to be emitted from the metal if the work function of the metal is 3.2 x 10-19 J
iv) Comment on this calculation with actual experimental data and conclude the validity of wave theory.